  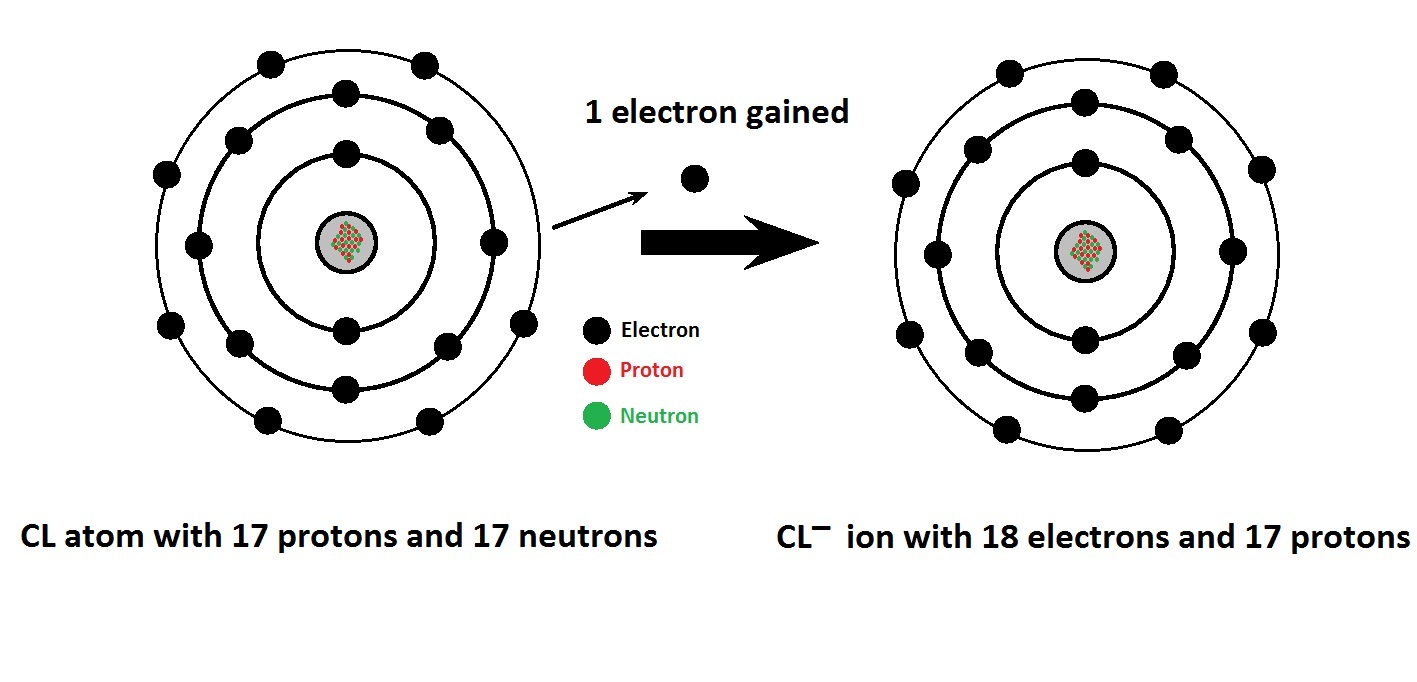
Cations are also called positive ions, and anions are also called negative ions. Anions are negatively-charged ions (meaning they have more electrons than protons due to having gained one or more electrons). ⚡️ Quick summaryĬations are positively-charged ions (atoms or groups of atoms that have more protons than electrons due to having lost one or more electrons). We use the word ion to refer to atoms that have a charge one way or another, and we use the words cation and anion to specify whether the charge is positive or negative. An atom becomes negatively charged if it gains extra electrons, and it becomes positively charged if it loses electrons. In an ordinary atom, the number of protons equals the number of electrons, so the atom normally has no electric charge one way or the other. The protons in an atom have a positive charge, the electrons have a negative charge, and the neutrons have zero charge. Electric charge is a basic property of elementary particles of matter ( protons, electrons, and neutrons). Anions have a negative charge, and cations have a positive charge.īefore we talk about what exactly that means (and why it matters), let’s talk about what we mean by charge. The difference is in the electrical charge. But seriously: anions and cations are both ions. A cation is a type of ion for cats (OK, fine, that’s not true, but it is pronounced ). An anion, pronounced, is a type of ion-an electrically charged atom or group of atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
